Solar-Driven Photocatalytic Seawater Splitting for Green Hydrogen Generation
Scientists have developed a new method to produce green hydrogen directly from seawater using sunlight, offering a promising pathway for clean and sustainable energy.
03 min reading
Three principal chemical technologies are used for CO₂ capture: amine-based absorption, calcium looping, and solid adsorption. Amine-based absorption employs liquid amine solvents to selectively remove CO₂ from flue gas, followed by thermal regeneration to release CO₂ and recycle the solvent. Calcium looping captures CO₂ via carbonation of calcium oxide (CaO) to calcium carbonate (CaCO₃), with high-temperature calcination regenerating CaO and releasing CO₂. Solid adsorption utilizes porous sorbents to selectively adsorb CO₂ from gas streams, with regeneration achieved through temperature or pressure swing processes. These technologies constitute key pathways for large-scale CO₂ capture and climate mitigation.
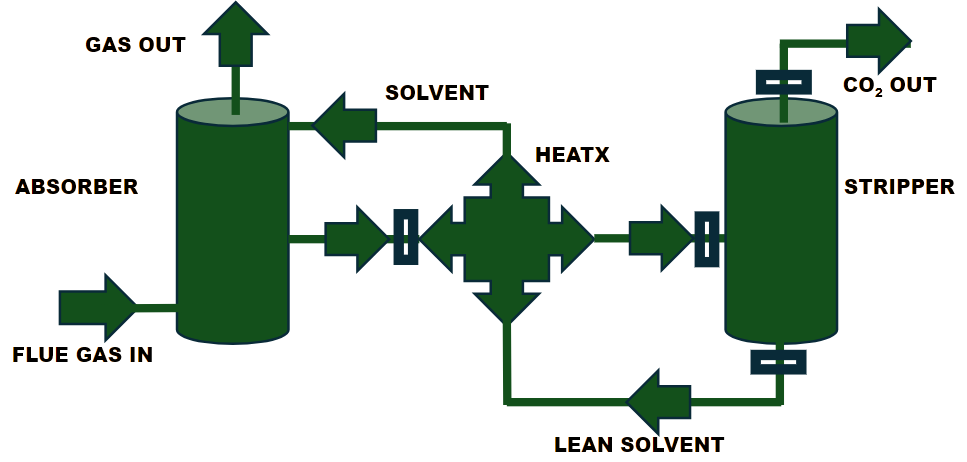
This is a post-combustion carbon capture technology in which an amine solvent is used to absorb CO₂ from flue gas. The absorbed CO₂ in the absorber column is then released (desorbed) in the stripper using high energy (temperature). The regenerated CO₂-free amine solvent (lean amine) is recycled back to the absorber. The captured CO₂ is then compressed and reused for other applications. Although this is a mature and highly efficient technology, it is expensive due to the high energy required for solvent regeneration.1
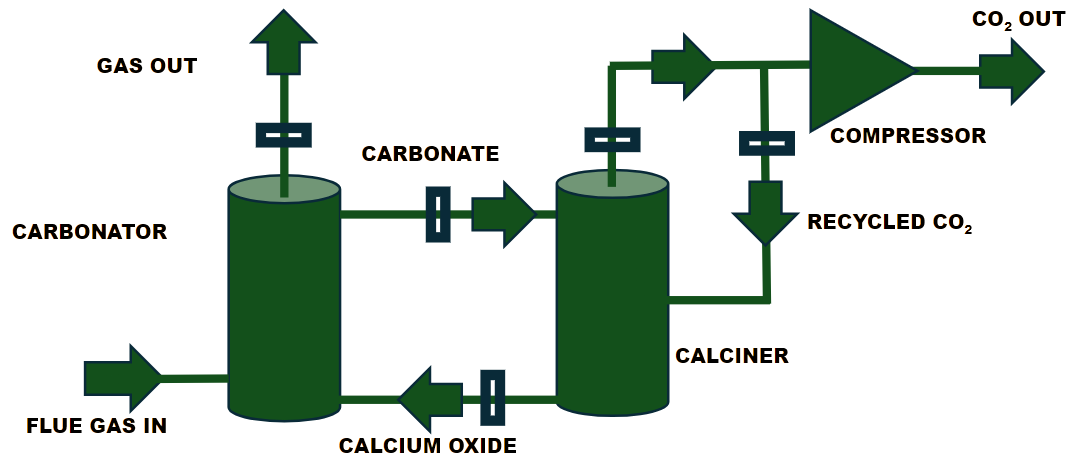
It is a promising technology for capturing CO₂ from high-temperature industrial sources as well as for direct air capture from the atmosphere. In this process, calcium oxide (CaO) in the absorber column reacts with CO₂ to form calcium carbonate (CaCO₃, or limestone). This reaction is reversible: in the stripper (or calciner) column, the limestone is heated to high temperatures to release CO₂ and regenerate CaO. The regenerated CaO is then reused to capture CO₂ again, completing the cyclic process.2
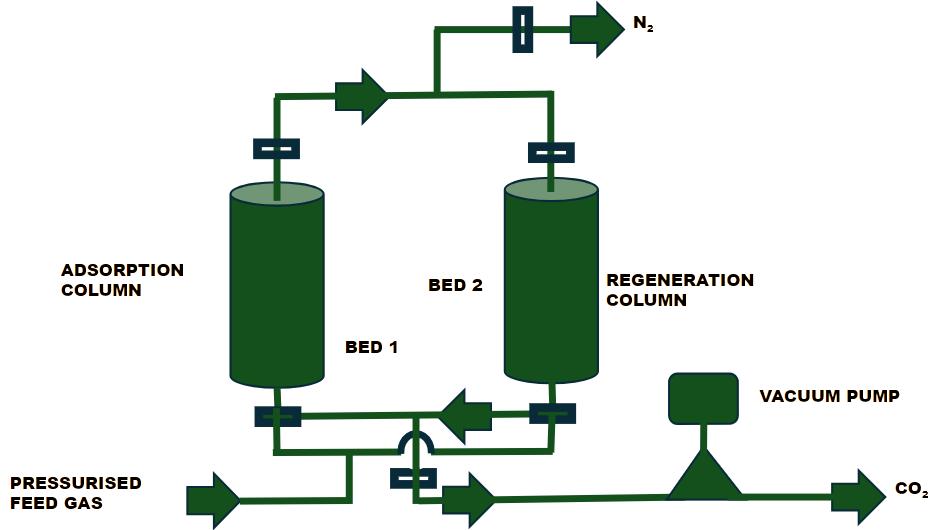
This technology is based on solid adsorbents that can selectively capture CO₂. As the feed air flows through a packed bed, CO₂ is adsorbed onto the sorbent surface. A combination of mild heating and reduced pressure is then applied to desorb the CO₂, making this a scalable and sustainable method for removing CO₂ from the atmosphere.3
Scientists have developed a new method to produce green hydrogen directly from seawater using sunlight, offering a promising pathway for clean and sustainable energy.
Novel electrochemical device separates electrode environments to accelerate CO₂ uptake while reducing energy consumption, paving the way for scalable carbon capture solutions.
Japanese scientists at the Institute of Science, Meguro in Tokyo have discovered a new type of liquid that could make capturing carbon dioxide from factories and power plants faster and more efficient.